Winkelwagen
U heeft geen artikelen in uw winkelwagen
Lever’s Textbook of Dermatology and Mckee’s Pathology of Skin have emphasized that the histological findings of an exanthematous drug eruption are often indistinguishable from those of viral exanthems, although the presence of eosinophils may favor a drug reaction [9,10]. Ackerman also believed that “drugs can elicit any of the nine basic patterns of inflammatory diseases in the skin, and none of those patterns is specific for a drug eruption” [11]. Nonetheless, various studies have highlighted certain histopathological pointers for both drug- and viral-induced exanthema, albeit they may not be pathognomonic.
Seitz et al. in their study on 91 patients of drug-induced exanthema described the most common reaction pattern to be the combined type of spongiotic and interface pattern, followed by the perivascular (15/26), spongiotic (10/13), vacuolar interface (6/10) and lichenoid interface patterns (4/5). Most importantly, no specific reaction pattern could be significantly attributed to drug-induced or non-drug-induced exanthem. Parakeratosis was present in a slightly but not significantly higher proportion of non-drug-induced exanthem than drug-induced exanthem. No significant differences in occurrence of dyskeratotic keratinocytes, extent of lymphocyte exocytosis or density of the dermal lymphocytic infiltrate were observed. Moreover, there was no significant difference in the presence of papillary dermal edema, accompanying vasculitis or extravasation of erythrocytes. A comparison of drug-induced exanthema and non-drug-induced exanthem did not yield a significant difference in the number of eosinophils within the dermal infiltrate. The results regarding the sensitivity (62.9%), specificity (41.1%) and positive (40.7%) and negative predictive values (69.7%) of a correct diagnosis showed that no valid conclusions can be drawn from histologic evaluations of skin biopsy specimens [12].
However, other authors have noted certain distinctive findings that may point towards either a drug-induced or a viral exanthem [ Figure 3 and Figure 4 ]. We will now highlight the important differences proceeding from the epidermis to the dermis.
The clinical manifestations of the SARS-CoV-2 infection are mainly respiratory, but also with multi-organ damage in variable proportions. In a study of 138 patients hospitalized in Wuhan the clinical features identified at onset were: 99% fever (not as common in other studies), 70% fatigue, 59% dry cough, 40% anorexia, 35% myalgias, 31% dyspnea, 27% sputum production. [9] In a study of over 5000 patients in New York, only 31% had a temperature of over 38°C at presentation. [10] Anosmia and dysgeusia have been identified as common symptoms in COVID-19. In a survey of 202 outpatients from Italy 64% reported alteration of smell or taste. [11] Gastrointestinal symptoms (abdominal pain, nausea/vomiting, or diarrhea) were found in 18% of the patients in a systematic review. [12]
In our case, the initial symptoms were predominantly respiratory, then digestive (diarrhea), neurological (anosmia), without any other organ dysfunction. Mild lymphopenia was noted but the neutrophils to lymphocytes ratio of 2.789 was interpreted as a favorable prognostic factor. A neutrophils to lymphocytes ratio of over 3.3 is cited in literature as a negative prognostic factor. [13] Using the online method suggested by Liang et al for calculating the risk of progression of SARS-CoV-2 infection towards severe disease, we obtained a moderate risk in our case. [14]
During the evolution of the case atypical cutaneous manifestations occurred, with rapid dissemination of an erythematous maculopapular, purpuric-like, mildly pruritic rash.
Exanthems are disseminated erythematous eruptions caused by medications (antibiotics, anticonvulsivants, tuberculostatics, antihypertensives, etc.), infections (respiratory viruses, enteroviruses, parvoviruses, herpesviruses, HIV, etc.), toxins released by some microorganisms (like staphylococcal scalded skin syndrome) or by autoimmune disease. [15,16]
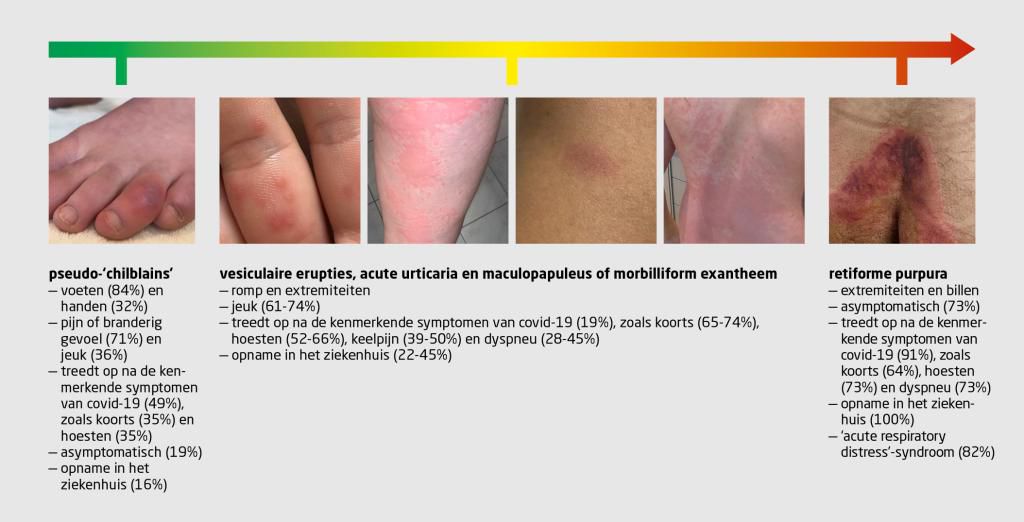
Viral exanthems secondary to SARS-CoV-2 infection are rarely described in literature. The first dermatological manifestations described in the context of SARS-CoV-2 infection were chilblain lesions on the plantar extremities and livedo reticularis. During the last months more types of cutaneous lesions were described in infected patients: urticarial, chickenpox-like, [17] vasculitic, morbilliform, [18] etc. A meta-analysis of 19 studies published by Sachdeva et al, showed that cutaneous manifestations can precede respiratory ones by 3 days (12.50% of cases) or can appear after 13 days (69.40%) from the onset of respiratory manifestations in SARS-CoV-2. [8] The medium interval for the appearance of cutaneous lesions was 9.2 days and the medium interval to remission was 8.7 days, according to a study of 132 COVID 19 positive patients with chilblain and erythema multiforme-like lesions, published by Fernandez-Nieto et al. [19] In our case the exanthema appeared 15 days from the onset of symtpoms and remitted after 10 days. Although there was a procoagulation status in our case (slightly raised d-dimmers), there were no acro-ischemic or vasculitic lesions. The absence of vasculitic lesions in this case suggests the fact that the pathogenetic mecanism of the viral exanthema was an immune response to the viral nucleotides. We excluded a secondary drug reaction by the lack of “en cocarde” lesions, mucosal involvement and pruritus. In order to exclude other causes of viral exanthema we conducted serological tests (IgM for parvovirus B19, human herpesvirus-6, 7, and Epstein Barr virus) which were negative. The pharyngeal swab was negative for streptococcus and the mildly elevated inflammatory markers along with the absence of the specific enanthema effectively excluded scarlet fever. Particular about this case is the persistence of anosmia for 6 weeks after disease onset.

The clinical manifestations of the SARS-CoV-2 infection are mainly respiratory but the virus can cause a variety of symptoms. Dermatological findings are less well-characterized. Data is scarce on their timing, type and correlation with the immune response.
We present the case of SARS-CoV-2 infection in a previously healthy woman who presented with respiratory symptoms and developed anosmia, diarrhea, and an erythematous maculo-papular rash on day 15 from symptom onset.
The nasopharyngeal swab tested by real time PCR for COVID-19 was positive. We interpreted this as a viral exanthema likely caused by an immune response to SARS-CoV-2 nucleotides.
She was treated with Hydroxychloroquine, Azithromycin and Lopinavir/Ritonavir, and the rash with topical corticosteroids.
All symptoms resolved except for anosmia which persisted for 6 weeks. At the 4- and 6-weeks follow-up the IgG titers for SARS-CoV-2 were high.
We must consider that SARS-CoV-2 has a multi-organ tropism. In our case, the SARS-CoV-2 infection had lung, nasopharyngeal, neurological, digestive, and skin manifestations. Identifying the different manifestations is useful for understanding the extent of SARS-CoV-2 infection. We not only present a rare manifestation but also suggest that cutaneous manifestations may correlate with immunity.
Keywords: exanthema, SARS-CoV-2, anosmia, clinical manifestations, immune response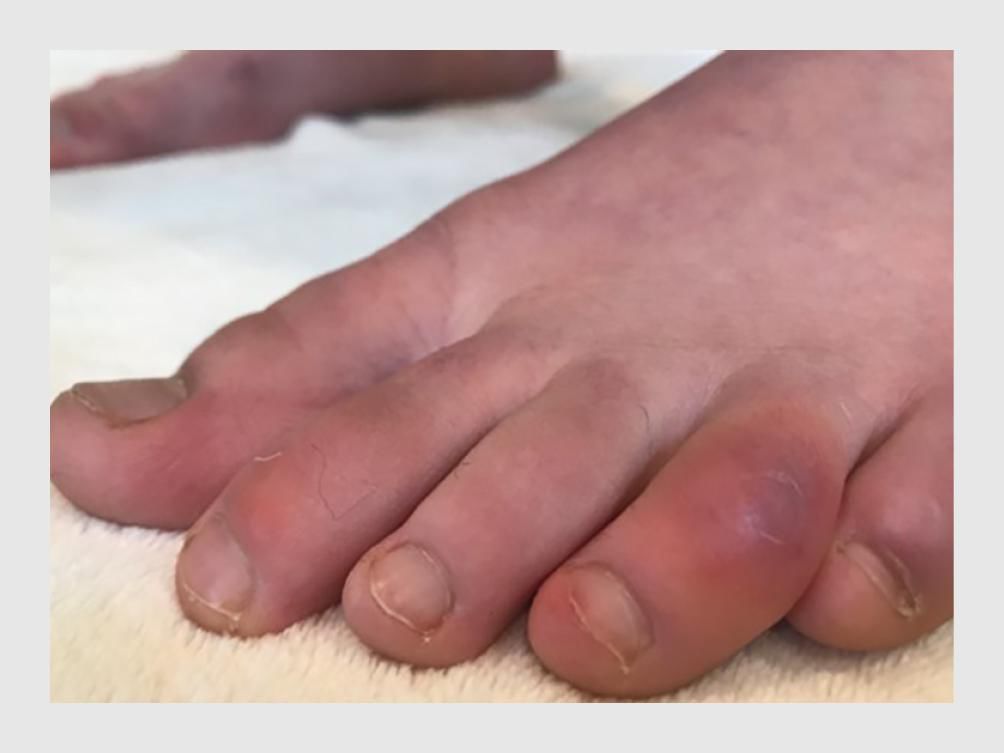
IL-5 and eotaxin cause activation and recruitment of eosinophils, thereby contributing to the development of skin inflammation in drug-induced maculopapular exanthems. Yawalkar et al. performed an IHC analysis of the dermal mononuclear infiltrate in drug exanthem biopsies and compared it with normal skin in control subjects. IL-5 expression was mainly found among mononuclear cells of the inflammatory infiltrate. Immunoreactivity for eotaxin, RANTES (regulated on activation, normal T cell expressed and secreted), IL-8 and to a lesser extent monocyte chemotactic protein-3 were also seen in the mononuclear cells. In addition, resident cells (i.e., endothelial cells and keratinocytes) also demonstrated positivity for eotaxin, RANTES and IL-8 [6].
The chronic dermal inflammatory infiltrate in a drug exanthem is primarily composed of CD3+ T cells. Within T-lymphocytes, CD4+ T cells are noted predominantly in the perivascular location, whereas equal proportions of CD4+ and CD8+ T cells are located at the dermoepidermal junction and in the epidermis [20]. Up to 20% of the infiltrating T cells in drug-induced maculopapular exanthem express perforin and granzyme B, which are important components of cell-mediated cytotoxic reaction. In addition to T cells, eosinophils are also present within the dermal infiltrate. These may also contribute to the generation of tissue damage via the release of various toxic granule proteins, such as eosinophilic cationic protein, major basic protein and eosinophil peroxidase.
In addition to drug exanthem, IHC may help confirm a suspicion of viral exanthem, e.g., the staining of skin biopsies with anti-measles virus (MeV) nucleoprotein and anti-MeV phosphoprotein can be of great value in confirming the diagnosis of this exanthem [15].